DO 604/32-1
| Period: | 2018-06-16 to 2020-07-30 |
|---|---|
| Partners: | Forschungszentrum Jülich, Goethe-Universität Frankfurt am Main, University of Pittsburgh |
| Funder: | German Research Foundation (DFG) |
| Project Manager: | Dr. Mario Rudolphi |
| Research Group: | High Temperature Materials |
Nearly all metallic construction materials rely on the formation of a thin and compact oxide layer as protection against the corrosive environment. The critical amount of chromium or aluminium required to form a protective oxide scale depends on the water vapour content in the atmosphere. A significant number of investigations have shown that the involved mechanism leading to this technologically important effect may be quite different for different alloys. Furthermore, at higher temperatures (around 900°C) the predominant mechanisms seem to be different from those prevailing at lower temperatures (around 600 °C). Recent results strongly indicate that hydrogen from the gas or from the reaction of water vapour with the (oxidized) metal surface influences the selective oxidation of the scale-forming element (e.g. Al or Cr). It is believed that this is caused by a direct influencing effect of hydrogen on the internal oxidation process and/or the oxide growth rate. This latter case (Fe-rich oxides on Fe-Cr-base materials and Ni-rich oxides on Ni-Cr(-Al)-base materials) might be due to hydrogen doping of the oxygen lattice or due to molecular transport of gaseous species (hydrogen or water vapour) through the oxide layer.
Up to now, a big deficit in elucidating the involved mechanisms has been the difficulty to detect hydrogen in a quantitative manner in the oxide scale and in the underlying internal oxidation zone.
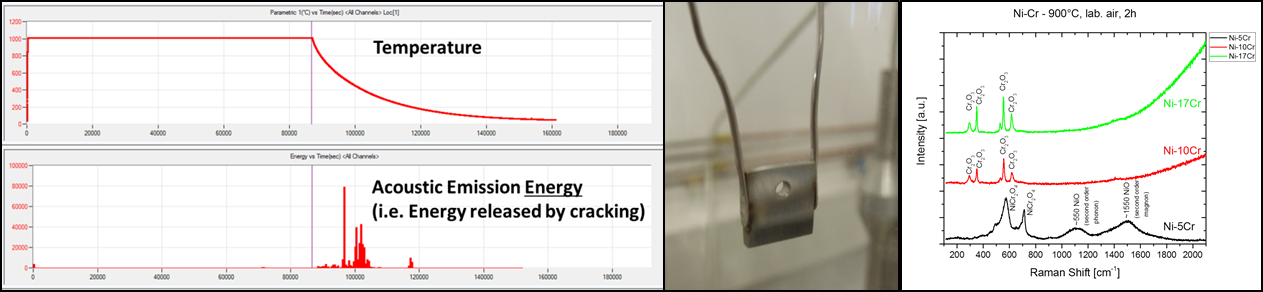
In this project the influence of water vapour on the formation of protective oxide scales is investigated. For this purpose, three model alloy systems are being investigated: FeCr, NiCr, and NiCrAl alloys. Microstructural investigations by SEM, EDX/WDX, TEM, and XRD are combined with the depth profiling techniques SNMS and GDOES and with in-situ determination of the oxidation kinetics by thermogravimetric methods. The main emphasis of the investigations is the depth-resolved detection of hydrogen using ion beam analytical methods. The techniques NRA and PIGE will be used in combination with well-defined sputter craters prepared by GDOES milling. In order to eliminate the influence of background hydrogen signals, some of the oxidation experiments will be carried out in D2- and/or D2O-containing (instead of H2- and/or H2O-containing) atmospheres. Acoustic emission measurement during high temperature exposure is used to detect microcrack formation and possible transport of molecular species along those crack paths. Finally, a model will be developed to extend the existing oxidation theory to the regime where hydrogen and/or water vapour will affect the formation of protective oxide scales.
back
Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) - DO 604/32-1