18904 N-1

| Period: | 01.01.2016 – 30.09.2019 |
|---|---|
| Partner: |
Universität Bayreuth, Chair of Physical Chemistry III, Research group Marschall Universität Ulm, Institute for Chemical Engineering, Research group Ziegenbalg |
| Funder: |
Federal Ministry for Economic Affairs and Energy (BMWi) via AiF Funding code: 18904 N-1 |
| Project Manager: | Julia Patzsch |
| Research Group: | Chemical Technology |
Heterogeneous photocatalysis has been known for a long time mainly for its applications in degradation of organic impurities and photocatalytic water splitting. However, especially in recent years, photocatalytic reactions have also become important in organic synthesis. The irradiation of semiconductors with light of a suitable wavelength produces very reactive states, so called electron-hole pairs, which can be used for a variety of chemical reactions such as oxidations, reductions or C-C couplings.
Using the example of quinoline synthesis (Figure 1) two important directions for the current development of photocatalysis are to be addressed in the present research project. On one side improved and optimized for organic synthesis photocatalysts will be developed and on the other side a scalable photoreactor concept in which the process can be operated continuously is aimed. The results should be applicable not only for the selected target reaction, but also for other heterogeneous photocatalytic reactions.
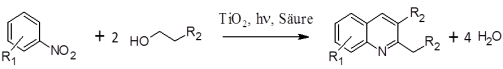
Figure 1: Photocatalytic quinoline synthesis from nitroaromatics and alcohols.
The work program is handled by three research partners:
At university of Bayreuth in the research group Marschall works on the further development of photocatalysts. The typically used titanium dioxide has a very high oxidation potential that can lead to undesirable side reactions and in some cases mineralize part of the substrate. In addition, it can only be excited by high-energy UVA light. Therefore, attempts should be made here to use photocatalysts based on other materials which have a significantly milder oxidation potential and can already be excited with visible light.
At DECHEMA Research Institute (DFI), kinetics studies are used to investigate and optimize the reaction behavior. Previous laboratory tests have achieved conversions of up to 54%. By varying stoichiometry, light intensity and temperature the reaction should be further optimized with regard to conversion and selectivity. The reaction is to be operated as a continuous process. In addition, the use of substituted educts is intended to further explore the spectrum of accessible products.
Photoreactor development is carried out at the research group Ziegenbalg, Institute for Chemical Engineering of the University of Ulm. Various photoreactor concepts are to be developed and investigated using rapid prototyping. In this case, both approaches in which the photocatalyst is suspended and those which are immobilized on the walls of the photoreactor are tested and compared.
back

Das IGF-Vorhaben Nr. 18904 N-1 der Forschungsvereinigung DECHEMA e.V., Theodor-Heuss-Allee 25, 60486 Frankfurt am Main wurde über die AiF im Rahmen des Programms zur Förderung der industriellen Gemeinschaftsforschung (IGF) vom Bundesministerium für Wirtschaft und Energie aufgrund eines Beschlusses des Deutschen Bundestages gefördert.
Dr. Jonathan Bloh
Tel.: +49 6172 89938-387
E-mail: jonathan.bloh
J. Patzsch, B. Berg, J. Z. Bloh, Front. Chem., 7 (2019), 289