LE 3727/1-1
| Period: | 2016-08-01 to 2020-05-31 |
|---|---|
| Funder: | Deutsche Forschungsgemeinschaft (DFG) |
| Project Manager: | Dr. Maren Lepple |
| Research Group: | High Temperature Materials |
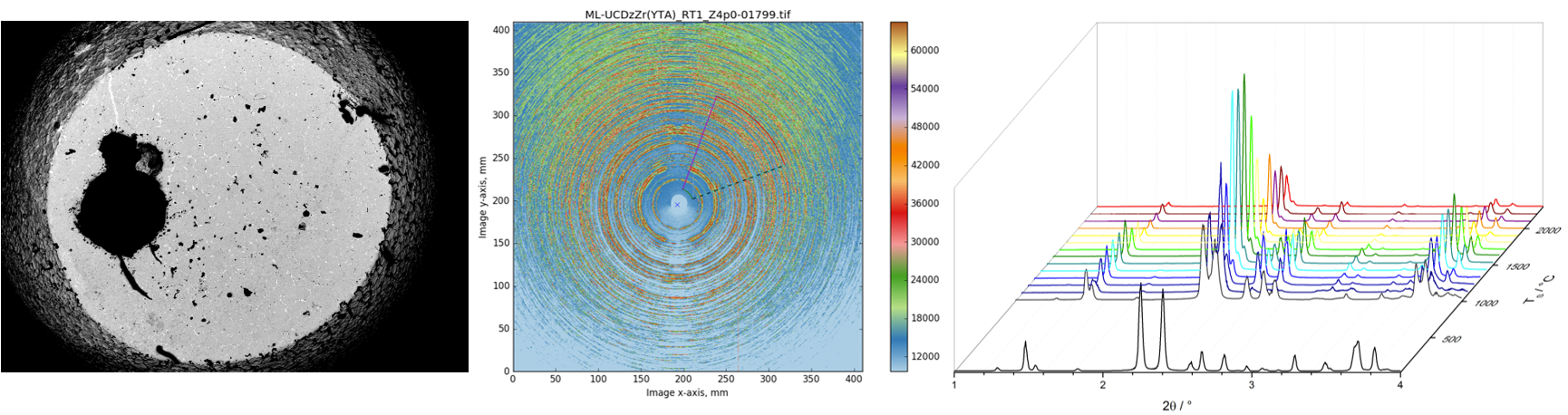
The efficiency of turbines can be improved by increasing their operating temperatures using enhanced thermal barrier coatings (TBCs). The ZrO2-Y2O3-Ta2O5 system is a promising materials system for future TBC applications because it exhibits an extended tetragonal phase region stable to high temperatures (~ 1500 °C) achieved by equimolar co-doping of zirconia with YO1.5 and TaO2.5. Compositions in the tetragonal phase fieldshow excellent properties relevant for TBC materials, such as low thermal conductivities, high toughness and resistance to hot corrosion. However, no systematic study in the tetragonal phase field has been performed to determine key coating compositions because the exploration of the complete composition range is a time-intensive undertaking. Therefore, advanced modeling and simulation methods need to be used to reduce development time by suggesting promising candidate compositions.
The aim of this project is to clarify the stabilization mechanisms of the tetragonal phase along the equimolar co-doped composition line in the ZrO2-Y2O3-Ta2O5 system by combining thermochemical investigations of phase stabilities with a comprehensive thermodynamic modeling approach based on accurate crystal structure information. Thereby a major contribution to the understanding and tailoring of TBC materials should be made.
The energetics will be investigated by using the CALPHAD approach. Key experiments will be combined with self-consistent thermodynamic modeling of the multi-component system. Enthalpies of formation of samples having different key compositions in the stable as well as metastable state will be derived from measured enthalpies of dissolution using high temperature oxide melt solution calorimetry. Phase stabilities, enthalpies of transformation as well as T0 temperatures will be determined using thermal analysis and will be connected to the stabilization effects. Furthermore, heat capacities of key compositions will be measured using differential scanning calorimetry as they are important for the understanding of thermal behavior of the compositions and are directly related to the temperature dependence of their Gibbs energies. The experimental data will be combined in the thermodynamic modeling of the multi-component system to develop a deep understanding of stabilization effects, phase relations and stabilities as well as constitution.
back

Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) - LE 3727/1-1
Dr. Mario Rudolphi
Tel.: +49 69 / 7564-492
E-mail: mario.rudolphi
M. Lepple, S.V. Ushakov, K. Lilova, C.A. Macauley, A.N. Fernandez, C.G. Levi, A. Navrotsky, Journal of the European Ceramic Society (2020)
M. Lepple, K. Lilova, C. Levi, A. Navrotsky, Journal of Materials Research 34 (2019), 3343