
| Period: | 01.02.2018 – 29.02.2020 |
|---|---|
| Partner: | PEUS-Testing GmbH |
| Funder: |
Federal Ministry of Economic Affairs and Energy (BMWi) via ZIM, Cooperation network: INET Funding code: 16KN074620 |
| Project manager: | Aneta Pashkova |
| Research group: | Chemical Technology |
Redox flow batteries (RFB) have one positive and one negative half cell separated by a membrane. The special feature of this battery type is the use of liquid tanks, which store the two different electrolytes. These are connected by tubings and pumps to the galvanic cell, in which the electron transfer takes place. The advantage of this design is that the power is scalable by the size of the electrodes and the storage capacity can be adjusted independently by the amount of stored liquid in the tanks. Due to these properties, RFB are particularly suitable for stationary energy storage, for example for the storage of decentralized solar power.
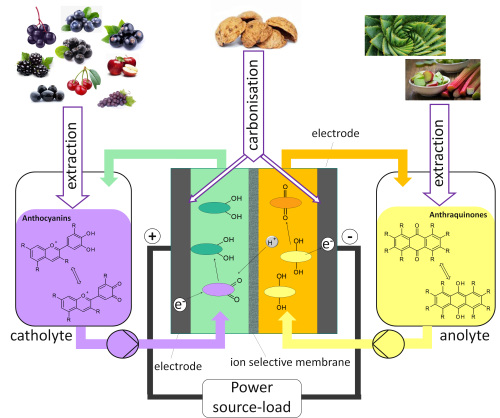
Schematical illustration of a bio-based redox-flow-battery out of sustainable resources
The present research project aimes at the thevelopment of a redox flow cell that uses a bio-based electrolyte instead of vanadium (V). Vanadium has the disadvantage to be an expensive metal, due to politically unstable mining countries and therefore insecure availability. The vanadium (V) of the positive half-cell is also corrosive, toxic and has a high overpotential, which is a challenge to the cell materials and one of the reasons for the low market share of the RFB.
The goal of the project is the development of a bio-based electrolyte for the positive half-cell of the RFB. Other aim is the cost reduction compared to Vanadium, as well as the development of a non-toxic and non-corrosive electrolyte with the same high electrical potential as V. Preliminary experiments from a feasibility study (BMBF project, code 031B0116) showed promising results with the substance group of flavonoids, more specifically with anthocyanins. These occur in large quantities in many fruits and serve as colorants and flavorings. They have antioxidant properties.
The BioFlow Cell will be developed in a cooperation between DECHEMA Research Institute (DFI) and PEUS Testing GmbH (PT). DFI focuses on the recovery and processing of the bio-based electrolyte, as well as its modification to the desired properties (e.g., long-term stability). The biologic substitute (flavonoids) should be extracted from by-product streams such as fruit pomace so as not to compete with the food industry.
Battery manufacturer PT is developing a cell technology optimized for the bioelectrolyte, i. Membrane development and surface / alloy development of the half-cell components, since these must not be oxidized by the electrolyte. The control technology and material management also require new developments compared to regular redox flow cells. One reason for this are the different voltages and the energy density of the electrolyte.
The negative half-cell can also be replaced by a biological alternative in the longer term. Here the substance group of anthraquinones shows great potential. However, synthetic anthraquinones are relatively inexpensive, so that the bio-based version can not easily achieve a cost advantage here. Therefore, the focus of this project is on the positive half-cell.
backDr. Jonathan Bloh
Tel.: +49 6172 89938-387
E-mail: jonathan.bloh
Dr. Aneta Pashkova
Tel.: +49 6172 89938-404
E-Mail: aneta.pashkova