IGF 19708 N
| Period: | 01.01.2018 - 31.03.2021 |
|---|---|
| Partner: | TU Braunschweig, Institut für Werkstoffe, Prof. Dr. J. Rösler |
| Funder: | AiF |
| Project Manager: | Dr. Stephan Lederer |
| Research Group: | Corrosion |
Aim of the Project
Titanium materials have been used in osteosynthesis and implant technology for about 30 years. Titanium and its alloys possess a favorable property profile: they combine high strength with a low elastic modulus, resulting in high bio-functionality, and thus minimizing the risk of implant failure. In addition, titanium alloys are biocompatible and corrosion resistant due to their native titanium oxide layer.
Currently the most common alloys in the field of medical technology are Ti Al6 V4 and Ti Al6 Nb7, which in addition to titanium contain aluminum and vanadium or niobium. However, if the oxide layer of an implant is damaged, harmful metal ions may be released and can enter the body and thus the bloodstream.
Due to the known negative effects of aluminum on the human organism and the cell toxic behavior of vanadium, an aluminum and vanadium-free titanium alloy is being developed at the Technical University of Braunschweig together with the DECHEMA research institute. In addition to titanium, this new alloys contain only elements that are already present in the human body or for which no negative effects are known. The newly developed alloys are required to meet and even exceed the mechanical properties of the standard material Ti Al6 V4.
For this purpose, first simulations are perfomed to identify suitable compositions which are then produced on laboratory scale and characterized in terms of their mechanical and chemical properties. In addition, extensive corrosion investigations are carried out in order to understand the release of metal ions and how they can penetrate into the implant-surrounding tissue. Promising alloys are produced on a larger scale, characterized and tested in real implant components.
In order to functionalize the material’s surface plasma electrolytic oxidation is carried out. This electrochemical process provides a plasma assisted conversion of the metal surface into a ceramic oxide layer. Consequently, the hardness and wear resistance are significantly increased and the material’s corrosion resistance is improved. Furthermore, biominerals, such as hydroxyapatite, can be deposited on the implant surface in order to improve the osseointegration of the implant
This research will provide medical technology companies with a new titanium material for the use in implants, characterized by an improved biocompatibility with comparable mechanical properties of currently in-use materials.
Results
At DECHEMA research institute, investigations are carried out on the corrosion and tribocorrosion behavior of the base alloy and the material surface is modified by means of plasma electrolytic oxidation.
Corrosion properties
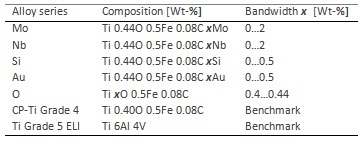
Titanium alloys of varying composition were examined for their corrosion properties (Table 1). The alloys were tested in 2 body-like fluids, Hank's solution + 0.1 M H2O2 and in artificial saliva + 2000 ppm fluoride, at 37 °C respectively. The benchmark systems were Ti-6Al-4V-ELI and Ti grade 4.
Table 1: Composition of the investigated Ti Grad 4+ alloys
The open circuit potential (OCP) of all alloys show a clear passivating behavior in Hank's solution at 37 °C. The OCP increases in the beginning from -600 mV vs. standard hydrogen electrode (SHE) to -500 mV to -450 mV vs. SHE after several hours. After about 12 hours, the OCP reaches stable values of -450 mV vs. SHE. The polarization curves show a clear passivation of the surface. Current densities up to 10 μAcm-2 at 1 V vs. NHE can be observed. As usual for titanium materials in chloride-containing media, a peak in the current density at approx. 2 V vs. NHE can be found, attributing to the residual oxidation of the titanium in the passive layer. No breakthrough in the range up to 4 V against NHE is visible.
There is a significant dependence of the corrosion resistance on the alloying elements. In general, alloying with Mo, Nb and Au improves the corrosion behavior in Hank's solution as well as in artificial saliva. Thus, the corrosion current density is reduced from 75 nAcm-2 for pure Ti grade 4 to approx. 15 nAcm-2 when adding 0.5 wt% Au. Alloys containing 2 wt% Mo or Nb, a decrease to 20-40 nAcm-2 is recorded. A similar effect can be observed in artificial saliva, however, the corrosion current densities are increased in general by about 2 orders of magnitude due to the slightly acidic pH of 5.5 and the addition of 2000 ppm fluoride. However, comparing pristine Ti grade 4 to novel alloys containing 2 wt% Mo or Nb, current densities are decreasing from about 1.8 μAcm-2 to 0.5 μAcm-2. On the contrary, alloying with Si exhibits a detrimental effect, i.e. a general deterioration of the corrosion resistance associated with an increase in corrosion current densities to about 80 nAcm-2 and 4 μAcm-2, in Hank’s solution and artificial saliva, respectively. The variation of the oxygen content showed no remarkable influence, probably due to the small range of 0.4 wt% to 0.44 wt% O.
Tribocorrosion
The tribocorrosion behaviour of the alloy depends on the load, the friction speed and the sliding distance. The wear volume increases with increasing load and distance. However, the wear volume decreases with increasing sliding speed due to less effective re-passivation and the associated lower oxidative wear. The influence of abrasive particles plays a major role in tribocorrosion and under dry conditions. The wear volume is significantly increased under dry conditions, which can be explained by the accumulation of abrasive third body particles in the wear track. After completion of the tribocorrosion experiments, a rapid re-passivation of the material can be detected in any case, which is useful for biomedical application.
Plasma electrolytic oxidation
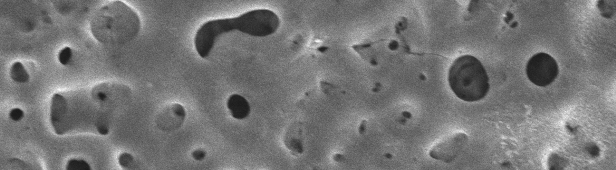
In order to generate bioactive hydroxyapatite-containing PEO layers, various Ca-P-containing electrolytes were used and the layer formation under DC and pulsed conditions was investigated. The morphology of the surfaces as well as metallographic cross sections of the generated oxide layers were characterized by SEM and EDX. The generated phases were analyzed with grazing incidence XRD. Layer thickness and roughness were determined for all layers.
In all the electrolytes used, hydroxyapatite-containing titanium dioxide layers with a thickness of several micrometers and a porous structure could be produced. Depending on the electrolyte used and the electrical parameters, the layer properties such as layer thickness, porosity and the phases formed can be varied. Overall, the PEO treatment significantly improves the corrosion resistance of CP-Ti alloys.
For the production of wear-resistant PEO layers, basic aluminate- and zirconium-containing electrolytes were used and the influence of relevant process parameters such as current density, frequency and duty cycle was determined. For this purpose, the PEO layers were tested after the PEO treatment under constant conditions in the tribometer. It turns out that the PEO layers show improved wear properties compared to the base material. The main effects for producing a wear resistant layer, are current density and frequency as well as interactions of higher order (especially current density*frequency). Furthermore, the morphology of the surfaces as well as metallographic cross sections of the generated oxide layers were characterized by means of SEM/EDX. The generated phases were analyzed under grazing incidence XRD.
In addition to crystalline titanium oxide (rutile and anatase), the formation of m-ZrO2 (baddeleyite) and α-Al2O3 (corundum) can be observed as well leading to the enormous increase in hardness and wear resistance of the PEO layers. Depending on the electrical parameters, the phase composition and the layer thickness can be varied over a wide range. Overall, the PEO treatment significantly improves the wear resistance of CP-Ti alloys.
back

Das IGF-Vorhaben Nr. IGF 19708 N der Forschungsvereinigung DECHEMA e.V., Theodor-Heuss-Allee 25, 60486 Frankfurt am Main wurde über die AiF im Rahmen des Programms zur Förderung der industriellen Gemeinschaftsforschung (IGF) vom Bundesministerium für Wirtschaft und Energie aufgrund eines Beschlusses des Deutschen Bundestages gefördert.