A. Solimani, T.M. Meißner, C. Oskay, M.C. Galetz
Solar Energy Materials and Solar Cells 231 (2021), 111312, DOI: 10.1016/j.solmat.2021.111312
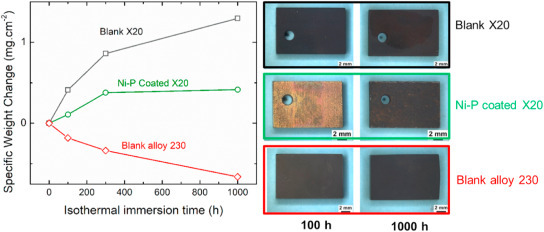
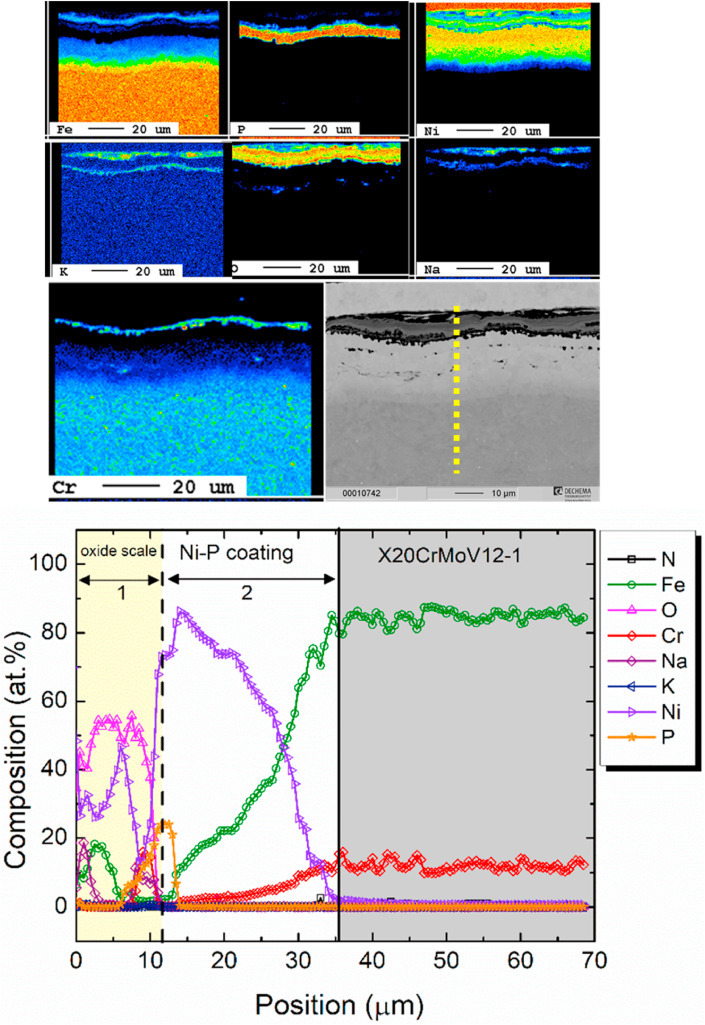
The corrosion behavior of an electroless Ni–P coating on low-Cr X20CrMoV12-1 steel as a cost-efficient substrate-coating combination is investigated in molten NaNO3–KNO3 nitrates at 600 °C. Results indicated that the as-deposited Ni–P coating was transformed to a thermally treated diffusion coating upon exposure to the molten salt without additional heat treatment prior to the exposure. Detailed evolution of the Ni–P coating and the oxidation behavior during the 1000-h immersion test is discussed. Results showed competitive corrosion resistance of the coating system compared to that of the sophisticated Ni alloy 230, with the advantage of an inherent barrier against Cr dissolution form the substrate. The significant corrosion resistance of the electroless plated Ni–P coating is attributed in part to the protective Ni--rich oxide scale and in other part to the in-situ formed Ni3P barrier at the oxide-substrate interphase boundary.