F. Lanoy, E.M.H. White, B. Schäfer, C. Tang, C. Schroer, M.T. Duerrschnabel, B. Gorr, M.C. Galetz
High Temperature Corrosion of Materials 103 (2026), DOI: 10.1007/s11085-025-10365-z
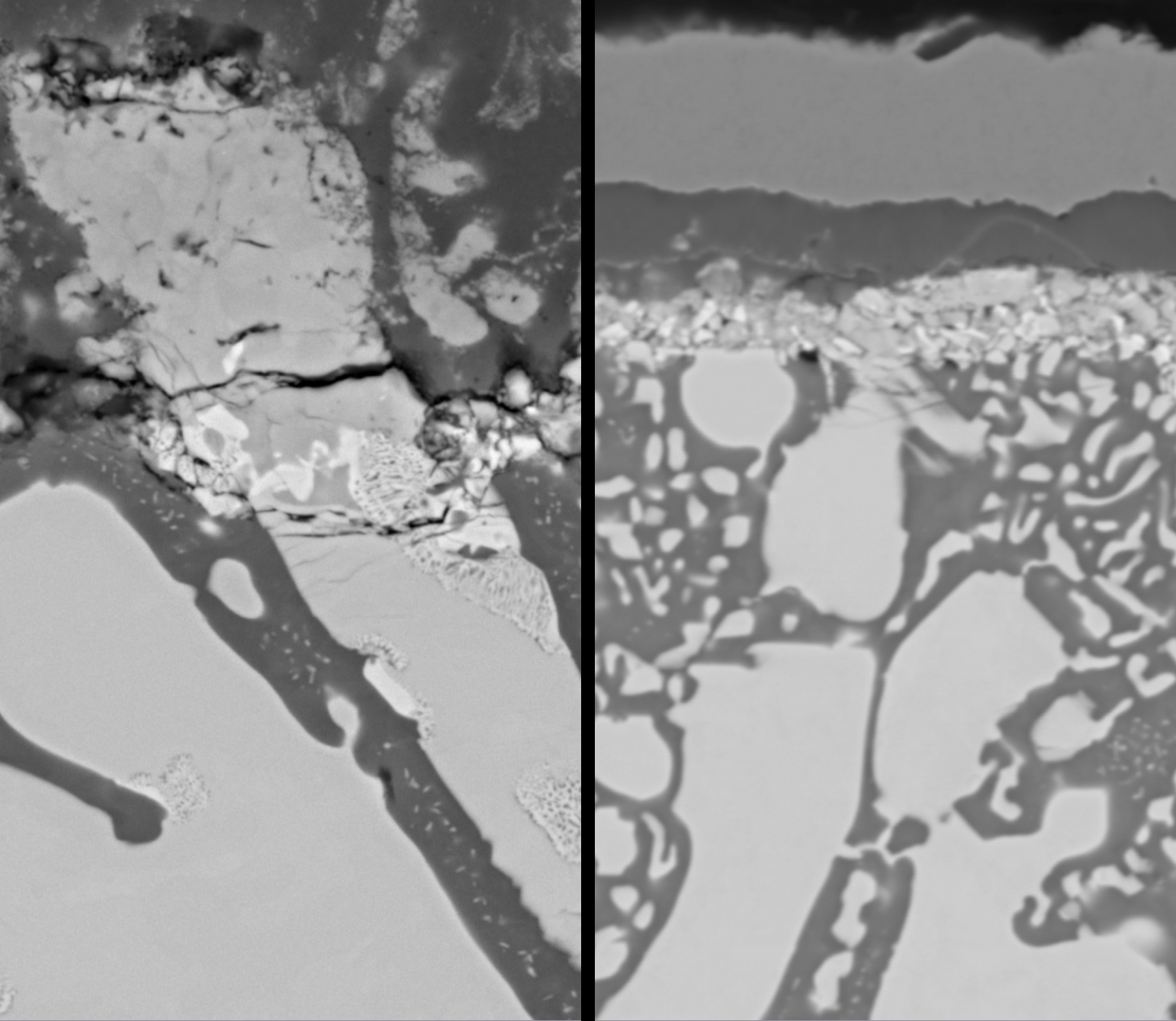
This study investigates the oxidation behavior of a binary Cr-20 at.%Ta alloy in a nitrogen-free, reduced oxygen partial pressure environment at 1000 °C for 48 h, aiming to clarify the intrinsic formation and growth mechanisms of protective (Cr, Ta)O2 oxides. Chromium outward diffusion primarily governs oxidation, leading to a duplex scale with an outer Cr2O3 layer and an inner (Cr,Ta)O2 subscale. Two distinct (Cr, Ta)O2 phases were identified: CrTaO4 (rutile structure) in outer regions and CrTa2O6 (trirutile structure) closer to the substrate, with CrTa2O6 confirmed as thermodynamically more stable through post-oxidation heat treatment and calculations. Thermogravimetric analysis revealed the parabolic oxidation constant of Cr-20 at.%Ta for fine-grained samples was eight times lower than pure chromium, highlighting the beneficial effect of the (Cr, Ta)O2 layer. The microstructure significantly influences the protectiveness: fine-grained alloys promoted a continuous (Cr, Ta)O2 layer, leading to enhanced oxidation resistance, particularly after a transient period required for the protective subscale to establish. This research underscores the critical role of (Cr, Ta)O2 and microstructure in developing advanced oxidation-resistant refractory alloys.